The Role of Rapid Toxicity Testing in Security Breach Evaluations
By Dan Kroll
There is little doubt that our water supplies are vulnerable to intentional contamination. The heightened sense of the need for security in the water industry since 9/11 has resulted in the need to respond to and investigate potential contamination incidents on a regular basis.
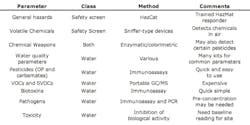
Depending on the situation a responder may or may not wish to perform more advanced field-testing on a given site. The EPA recommends a variety of possible tests that may be included in expanded field-testing. See table 1.
There are a wide variety of tools, kits and instruments that could be readily adaptable to expanded field-testing.
Many of these methods are new and unproven, and caution should be the rule when initially using them until confidence has been built-up as to their ability to provide accurate results. The EPA runs a program, known as the Environmental Technology Verification program (ETV), which independently evaluates such technologies. Evaluation by the ETV program doesn't certify or endorse the verified products but simply provides an independent source of data as to their performance as they relate to the specified test conditions. A description of the ETV program and the technologies that have been verified can be found on the ETV website. One of the technologies that offers wide applicability and the ability to respond to a variety of potential contaminants is toxicity testing.
Toxicity Tests
Toxicity is the ability of a chemical or mixture of chemicals to cause a living organism or biological system to undergo adverse effects upon exposure. These effects can include negative impacts on survival, growth, reproduction, etc. Toxicity tests are laboratory experiments, which attempt to detect or quantify toxicity in a sample by measuring the results that exposure produces on standard test organisms.
From the security standpoint there has been a number of on-line toxicity monitors developed and deployed utilizing organisms such as muscles and fish. This article will deal only with toxicity methodologies that are amenable to field deployment and that could be useful in on-site testing of a potential attack.
There are a variety of methods available for field toxicity testing. They rely on a number of different organisms and methodologies to determine the relative toxicity of a water sample. One thing to keep in mind with toxicity testing is that it is a relative measure and can vary considerably from location to location. Knowledge of the specific location's baseline toxicity is imperative when looking for significant deviations. It is also important to be cognizant of the fact that the toxicity of a water source being used as a blank can vary. This includes deionized water. In the course of the deionization process the resins can begin to release materials that can cause a change in toxicity. Therefore, an apparent change in your samples toxicity may in fact be due to a change in your blank standards toxicity. It can be a good idea to maintain a large supply of the baseline water against which all future testing is performed.
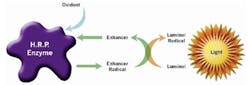
Chemiluminescence: One method of monitoring toxicity is based upon chemiluminescence. This method is the one used in the Severn Trent Services EcloxTM kit. The reaction of luminal and an oxidant in the presence of horseradish peroxidase, which results in the chemical production of light or chemiluminescence, can be used to detect the presence of toxins. Any free radical scavengers or anti-oxidants such as those contained in feces or urine will interfere with the reaction thus reducing the light emission. Substances such as phenols, amines, heavy metals or compounds that attack or coat the enzyme will also reduce the light output. The light output is plotted over time and produces characteristic curves. Results are compared to de-ionized water (DI). Samples containing pollution will give lower light levels. See Figure 2.
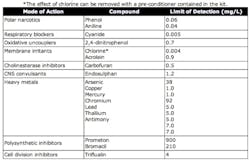
The Eclox™ method is extremely robust and has been hardened for field use by the British military. This method has been shown to be effective against a wide variety of substances that could be potential risks. See Table 2. As the kit is commercially available it comes co-packaged with a variety of subsidiary tests. These include methods for pH, conductivity, color, arsenic and cholinesterase inhibiting substances including pesticides and nerve agents.
Bioluminescence: Another way to monitor toxicity is based on the production of light by various species of luminous bacteria. CheckLight LTD from Israel has developed a system based on inhibition of light production from the bacteria Photobacterium leiognathi. This system has the ability to use different buffer screening systems to differentiate between organic pollutants and metal toxins. More common is the use of the luminescent bacteria Vibrio fischeri. This bacteria is the basis of the BioToxTM Flash Test produced by Hidex Oy of Finland and the MicroTox® and DeltaTox® test kits produced by Strategic Diagnostics. These kits have been shown to be capable of responding to a wide variety of different compounds. They all utilize the reduction of light output as measured by a luminometer, a fairly expensive piece of equipment, to detect toxicity to the bacterial cultures. There may also be some problems with removal of chlorine in finished drinking water samples, which can skew results.
Bacterial respiration: Another method for measuring toxicity is the inhibition of bacterial respiration. PolyTox™ sold by InterLab Supply uses standard dissolved oxygen electrodes to measure respiration of a specially formulated bacterial culture that is sold under the PolyTox™ brand. Oxygen consumption is a good measurement of overall bacterial health; however this method does present some problems. Drawbacks are that the test requires the use of a dissolved oxygen electrode, which is a touchy piece of equipment. The samples must be aerated for 30 minutes before staring the test. This aeration could result in the loss of volatile toxins from the sample. Finally, only one test can be run at a time; multiple tests require long periods of time coupled with extensive sample manipulation.
Another method based on bacterial respiration is the ToxTrak™ Rapid Toxicity testing system produced by Hach Co. Rather than directly measuring oxygen consumption the ToxTrak™ system utilizes a colorimetric system based on the rate of reduction of Resazurin dye. As the bacteria actively metabolize, the dye is reduced from blue to pink. This is normally a slow process, but the Hach system makes use of a patented accelerator solution that increases the rate of reaction and allows the test to be completed in as little as 45 minutes. Inhibition of the rate change is indicative of the presence of toxicity. The color change can be measured with any spectrophotometer or colorimeter capable of measuring at 600-610 nm. The color change is actually capable of being measured visually and a color disk comparator method is available. This system is very inexpensive and has been shown to react to a wide variety of toxins. While virtually any bacteria may be used in the test, one drawback is that cultures must be grown and maintained in advance of testing.
Testing with Daphnia and other Invertebrates: The utilization of invertebrates such as the water flea Daphnia has long been a mainstay of the toxicology laboratory. Strategic Diagnostics offers a variety of test kits in their MicorBioTest line. These tests make use of a variety of different organisms including: Tetrahymena thermophila, Brachionus calyciflorus, Brachionus plicatilis, Thamnocephalus platyurus and several Daphnia species. Toxicity in many of these tests is judged by the ingestion or failure to ingest red microspheres that are clearly visible in the organism's digestive tract. This is a much easier endpoint to judge than traditional assays that look for lethality or changes in behavior.
Aqua Survey Inc. of Flemington, NJ under the brand name IQ Toxicity Test provides another simplification of classic invertebrate toxicity testing. This method is based on fluorescent tagging of a sugar molecule that is placed in the Daphnia's food. If the Daphnia are happy and actively metabolizing they will ingest the sugar and cleave the molecule. This cleaving of the molecule causes the organisms to become fluorescent under UV light. It is basically a variation of the classic MUG test for E. coli bacteria. If the Daphnia glow, there is no toxicity. If they don't, there is. Daphnia testing is extremely sensitive and may actually be overly sensitive to common drinking water constituents in some cases.
The largest draw back to all invertebrate testing is culture maintenance. For emergency testing, a usable culture needs to be maintained at all times. In some cases organisms in a specific stage of development or state of hunger are needed. This is hard to maintain for an emergency program and is probably better suited to an ongoing program of laboratory testing where the organisms are more easily maintained in the proper state.
All toxicity-testing methods require knowledge of a base line. Also, some toxicity methods may be too sensitive when testing in the distribution system. Water treatment chemicals or simply common constituents of drinking water -- such as trace metals that are not toxic to humans -- may adversely affect them. The choice of whether or not to use toxicity methods depends a lot on the resources available for such testing. A dedicated program that maintains some knowledge of baseline conditions can take a sizeable amount of resources. Also, due to the differences in response to various toxins from different toxicity methods (see table 4), it is often a good idea to utilize more than one method or back them up with subsidiary tests such as the ones provided in the Eclox™ kit. If proper techniques, an understanding of their limitations and knowledge of normal conditions are utilized, rapid toxicity testing can be a valuable tool in the forensic investigation of water security breeches.
WaterWorld Online, July 2010