Bottling a Winning Clean-in-Place Solution
Vast amounts of water are used in the beverage bottling process, especially in Latin America which hosts some of the world's largest facilities. Beth Boeh and Juan Carlos Christy discuss the use of electro-chemically activated clean-in-place methods to help reduce the use of caustic and toxic chemicals.
The beverages we drink every day – many from familiar giants like the Coca Cola Company, Pepsi, and Dr. Pepper Snapple Group – typically arrive in our hand through the bottlers, independent companies or franchisees who buy the syrups and concentrates from the big names, then mix, bottle, package and distribute the finished products.
Beverage bottling is particularly big business in the high-growth Latin American market. Coca-Cola FEMSA, the largest independent bottler in the world serving the Latin American market, accounts for one in ten Coca Cola products sold globally, delivering more than two billion unit cases each year. With one plant producing a staggering 14 million bottles each day – almost 10,000 bottles per minute - every second on the production floor counts.
In the carbonated beverage bottling process, scrupulously high standards for sanitation is a must. The brands we grew up with have depended on the perfection of their product for generations. A clean-in-place (CIP) process is used in many beverage bottling and canning plants to clean preparation tanks and lines that are used for sugars, syrup and water during the beverage mixing process. Stringent cleaning prevents flavor carryover, controls microbial contamination and eliminates organic residues like precipitated proteins, carbohydrates, and minerals that potentially harbor bacteria.
Beyond contamination of the product, bacteria can lead to microbial induced corrosion (MIC) if biofilms are allowed to form in the production system. MIC can be particularly damaging to beverage production equipment.
The CIP cleaning process is, like it sounds, largely done in-situ, where the vast majority of piping, tanks, pumps and other components are left in place, not disassembled during interior cleaning. CIP cycles of a single beverage line can occur multiple times per day, depending on the production process used for a given line. Compared to dismantling systems in traditional cleaning, the CIP cleaning method offers a rapid, labor saving cleaning method that is more reliable and reproducible as compared to traditional cleaning approaches.
While the CIP process is a relatively short-cycle process, it can still be one of the more time-consuming aspects of bottling plant operation and maintenance. Traditional CIP cycles can take 90 minutes or more to complete, resulting in substantial down time for a production line if multiple CIP cycles are required on a daily basis. Time lost during line cleaning – even CIP - can translate into three to eight non-productive hours per plant per day. Many traditional CIP methods require that all or some of the steps of the CIP cycle be heated, incurring additional production loss through extended downtime as well as significant energy costs.
Finally, vast amounts of water is used during CIP, and this water usage is problematic as beverage manufacturers attempt to decrease the overall environmental footprint of their production facilities. As a result, many beverage producers are constantly seeking to reduce their CIP cycle times without sacrificing sterility, safety, or adding additional cost.
In Latin America, some beverage bottling plants are using an electro-chemically activated (ECA) clean-in-place method to save time and money. On-site, on-demand generation of a sanitizing oxidant – disinfectant - is ideal for CIP beverage processing, reducing costs by increasing efficiency and therefore, valuable beverage production time. The ECA cleaning solution is a single-component solution that replaces traditional four and five step CIP processes with a rapid three step cold-CIP process: rinse, treatment, and final rinse. Using a non-thermal technology reduces energy consumption, the rapid cleaning cycle significantly improves beverage facility production rates, and the water used to accomplish CIP is substantially reduced.
The process
Based on decades-old scientific principles, on-site ECA generators apply electricity to a solution of salt and water, which produces chlorine and other oxidant species. ECA generators have been used to treat municipal water to U.S. Environmental Protection Agency (EPA) drinking water standards for over 30 years and have a number of industrial applications - including providing regulatory approved disinfectants in the beverage process, power plants, and aquatics applications.
ECA generators produce chlorine when a solution of sodium chloride is passed through an electrolytic cell and electricity is added. Incoming water passes through an ion exchange water softener to remove calcium- a necessary step to prevent carbonate scale build up on the electrodes. Softened water feeds the electrolytic cell while a soft water sidestream fills a brine tank, which generates a concentrated salt solution. The near-saturated brine is then injected into the softened water stream entering the electrolytic cell.
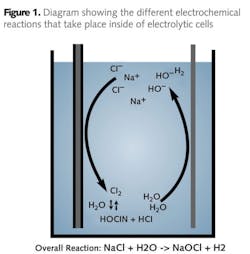
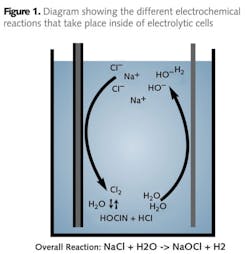
With the dilute salt solution inside the electrochemical cell, current passes through the cell, producing a strong oxidant solution. After exiting the electrolytic cell, the oxidant solution is stored in an oxidant tank. The electrolytic cell is the heart of the ECA generator, consisting of two electrodes, the anode and cathode, designed so that both make contact with the mixed water and brine solution. A voltage is applied to the cell so that current flows through the cell, causing chemical reactions to take place at the surfaces of both electrodes, producing the disinfectants. Oxidation reactions are carried out at the anode where two chloride ions (Cl-) are stripped of one electron each to produce chlorine which is dissolved in the solution.
The geometric, hydraulic and power configuration of the cell makes it possible to produce oxidants other than chlorine – the mixed oxidant solution - that can provide enhanced removal of microbiological contaminants. Chlorine production is balanced by the reduction reactions that occur at the cathode where water (H2O) is converted into hydroxide ions (OH-) and hydrogen gas (H2).
Hydrogen gas in the form of bubbles is produced during electrolysis at the cathode. Passive and active ventilation systems remove the gas from the generator and piping before it can enter the oxidant storage tanks, providing maximum system safety.
Why ECA Clean-in-Place for Beverage Processing?
Increased beverage production time using a three step ECA clean-in-place process can result in significant margin improvement and chemical cost savings, while the non-thermal cleaning method reduces energy consumption. Additionally, a three-step cold ECA CIP process can dramatically reduce the consumption of water. Because the only consumables are salt and electricity used to generate the chemical and regularly purchasing and shipping expensive chlorine chemicals is eliminated, ECA generators typically produce chlorine at a much lower cost than traditional delivery methods.
And since many beverage bottling plants will already have high-quality food-grade salt available, procurement of the only "chemical" required for the generator is simplified. Decreased costs associated with transportation and safety regulations can lower insurance premiums and provide additional savings.
Produced on-site, on demand, the ECA-generated mixed oxidant solution is an inherently safer disinfectant for the beverage industry, using non-toxic consumables - salt, water, and power - to generate the disinfectant. The produced solution has a relatively low concentration with moderate pH. Without hazardous chemicals transported, generated or stored, ECA offers superior safety for beverage plant personnel and the surrounding community.
Since the ECA-generated mixed oxidant solution is highly effective at eliminating algae and biofilm, bacterial contamination is reduced or eliminated and disinfectant requirements are reduced. Taste and odor carry-over are eliminated when switching lines from one flavored beverage to the next, plus the solution is easily rinsed from the system, eliminating residual "chlorine" taste and odor quickly.
Moreover, effective removal of microbiological contamination of process lines will prevent the occurrence of microbial induced corrosion resulting from biofilm formation. Compatibility of the solution with existing materials of construction (304 and 316 stainless steel, etc) are excellent and below industry standards for corrosion when the solutions are used at the appropriate strength.
Many bottling companies are taking a proactive approach to corporate environmental responsibility in partnership with the big-name beverage companies. In addition to the reduction in use and potential accidental release of hazardous chemicals, transportation of chemicals from factories to the beverage processing plant is dramatically reduced. Many purchased sanitizers are diluted in water to make handling safer, but the dilution water requires transportation, often significantly more weight and volume than the salt needed for ECA processes. This reduces a beverage processing plant's carbon footprint since less fossil fuel is needed to supply the plant with disinfectant. Additionally, the reduced CIP cycle time translates directly into a decrease in the overall use of water, resulting in a further reduction in the overall environmental footprint of the production facility.
Conclusion
Demand for bottled beverages is high with Mexico holding the soda-drinking record: an average consumption of 462 8-oz bottles per year per capita. Bottlers using on-site ECA clean in place technology to clean their lines are reducing costs, improving performance, and employing a safer, more environmentally sustainable method. By generating the sanitizer on site with common consumables - salt and water - caustic and toxic chemicals are eliminated, improving plant safety and environmental sustainability. The rapid treatment cycle saves time and money by increasing plant production. And when bottles are being filled at over 160 per second in one plant, well….every second counts.
Author's note:Beth Boeh, MIOX Corporation and Juan Carlos Christy, Quimiproductos. For more information, please email: [email protected].
More Water & WasteWater International Current Issue Articles
More Water & WasteWater International Archives Issue Articles