Facility Selects Magnesium Hydroxide as Alkalinity Supplement
When choosing an alkali for wastewater treatment, cost, ease of use, safe handling, effective pH control and long-lasting alkalinity should be factored in the selection process. Magnesium hydroxide slurry, an industrial form of "milk of magnesia," can be a competitive alternative, as staff at a municipal wastewater treatment plant on the Kentucky side of the Ohio River discovered in their investigation of alternative alkalis.
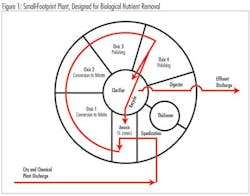
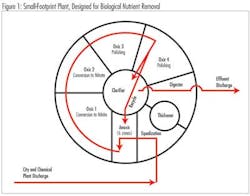
The municipality used soda ash (Na2CO3) for nearly 15 years to supplement alkalinity in the biological treatment of wastewater. The influent, which is comprised of 85% industrial waste and 15% municipal waste, is treated in oxic and anoxic zones to facilitate nitrification and denitrification.
Combined influent is pumped through four oxic zones via selectors. Alkalinity and oxygen demand are highest in oxic zone 1 where ammonium is converted to nitrite. Alkalinity is increased by supplementation. Diffusers in all the oxic zones roil the wastewater, increasing oxygen absorption through increased surface area. Further conversion to nitrate occurs in oxic zone 2. Denitrification occurs in the anoxic zones by recycle.
The operations staff expressed interest in investigating a conversion from soda ash to magnesium hydroxide to gain technical benefits and reduce operating costs. It was determined by the municipality early in the program that magnesium hydroxide treatment could provide an annual savings of more than $100,000 in chemical costs alone.
The basis for testing magnesium hydroxide was its improved alkalinity when compared to soda ash since one dry pound of soda ash is replaced by 0.56 pounds of magnesium hydroxide. Magnesium hydroxide also maintains a controlled pH even with an over- addition due to its buffering capability, which is critical in the state of Kentucky where an upper discharge limit of pH 9.0 cannot be exceeded. Additionally, operators at the facility expressed concerns about the long-term availability of soda ash as well as the cost of water spray to mitigate the oxic zone foam caused by soda ash.
Ease of handling, lower chemical consumption and therefore lower freight costs, and improved sludge quality were other benefits that would be investigated.
Following a successful plant trial that demonstrated the benefits of magnesium hydroxide, the treatment plant manager and all operations staff were in favor of an alkali conversion. Conversion capital costs were dramatically reduced by converting a dormant 8,000-gallon methanol tank to magnesium hydroxide storage. In June of 2007, the State of Kentucky and city council approved the process change and $38,000 for the conversion.
In October 2008, a water treatment consultant proposed another alternative alkali program that was projected to achieve an annual chemical cost savings of $370,000. In 2009, magnesium hydroxide was replaced with lime slurry, which was lower in cost but had a propensity for scaling and pH excursions. Since one dry pound of hydrated lime is equivalent to 0.79 pounds of magnesium hydroxide, an increase in alkali shipments and freight cost, and lower alkalinity contribution were expected.
After injecting lime slurry for 16 months in the biological treatment process, the city came to realize that the costs associated with lime's operational issues outweighed the chemical cost savings. The city's treatment plant experienced excessive scaling which led to flow interruptions, plugging of lines and pipe fittings, and equipment failures. The plant also experienced a large increase in solids at the belt filter presses requiring increased landfill disposal.
Some areas of the aeration basins had three feet of sludge collection on the bottom which jeopardized the effectiveness of the air diffusers. In addition, the plant increased its alkali shipments from 10 truckloads per month of magnesium hydroxide slurry to 20 - 30 truckloads per month of lime slurry.
The city requested a site visit from Martin Marietta Magnesia Specialties to discuss converting back to magnesium hydroxide and recommendations to improve magnesium hydroxide utilization. The city converted back to magnesium hydroxide in late 2010 and implemented the following changes based on Martin Marietta's recommendations:
- The upstream industrial discharger was to provide advance notification to the city of high ammonia, high COD, and high flow conditions to allow the city to make alkali adjustments.
- More sludge was to be recycled back to the process to use any unreacted magnesium hydroxide and determine if the alkali feed rates could be decreased.
The operations staff was pleased to see magnesium hydroxide returned to use as an alkalinity supplement for biological wastewater treatment, as it eliminated the operational issues associated with lime.