Disinfection Design, Operation Implications for Reuse Systems
By Edmund A. Kobylinski and Gary L. Hunter
Disinfection is the heart of water reuse systems. Unfortunately, designing for future flows can result in a chlorine contact basin that is too big at startup and remains so if anticipated reuse projects do not materialize. When development projects fizzle, utilities can be left with a large chlorine contact basin that has a retention time of not the desired two hours but one or two days. This excessive retention time can prevent facilities from meeting chlorine residual standards and from producing water that meets NPDES permit and reuse criteria.
Wastewater Treatment Plants (WWTPs) face multiple operating challenges with chlorine systems. A major challenge is how to maintain a residual chlorine concentration at the end of the basin with a retention time of two hours or more. Before nitrogen limits were established, most treatment plants used chloramine for disinfection. However, more facilities now disinfect wastewater with free chlorine, which tends to react with anything it can and cause formation of THMs that may be regulated. So as chlorine is consumed in side reactions, more chlorine must be added to comply with CT requirements. The question then becomes how to balance retention time, chlorine dose, disinfection, chlorine decay, and disinfection byproduct (DBP) formation with only one control variable.
Many believe that reuse does not impact the main WWTP, while in fact it does. Special disinfection design features and different operating strategies are needed to accommodate reuse projects.
Chlorine Chemistry
Development of an effective control strategy requires a thorough understanding of chlorine chemistry. Chlorine’s effectiveness as a disinfectant depends on its concentration and on contact time. An indiscriminate oxidant that reacts with a wide range of organic materials, chlorine will react with the first reducing agent with which it comes into contact, oxidizing the organic material in cells and killing the target organism. As organic compounds react with chlorine, they are usually partially oxidized, thereby producing a DBP. The four primary DBPs are subject to very restrictive water quality standard-based limits. This is where the dilemma begins: WWTPs must add a dose of chlorine that will simultaneously meet established disinfection goals and minimize DBP formation to comply with water quality-based permit limits.
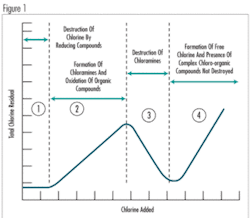
Breakpoint chlorination is a stepwise reaction that begins with the formation of monochloramine (see Figure 1, Zone 2). As more chlorine is added, all of the ammonia is instantaneously converted to monochloramine. As still more chlorine is added, dichloramine will begin to form, requiring about 20 minutes to proceed to completion (Zone 3). However, the presence of more-concentrated free chlorine causes more rapid formation of dichloramine, because the rate of dichloramine formation is a function of the residual concentration of free chlorine. If enough chlorine is added, dichloramine will be converted into trichloramine, which will cause chlorine residual to decline. Trichloramine formation is also a rapid reaction (Zone 3). Once all of the dichloramine is converted to trichloramine, the free chlorine concentration can increase (Zone 4). This relationship is very important in the selection of on-line chlorine residual analyzers for control of the disinfection process.
Breakpoint chlorination cannot be stopped, but it should be taken into account in developing a control scheme. Any chloramine left in solution when wastewater is dechlorinated will be converted back into ammonia.
Design Considerations
Another important question is how to maintain a free chlorine residual when a long retention time is required. Chlorine will react with anything it can, so maintaining a CT value greater than 450 mg/L/min in a 90-minute retention time requires maintaining a chlorine residual of at least 5 mg/L – which requires a higher chlorine dose because of side-reactions and decay. At this higher initial chlorine concentration, unwanted side reactions occur, requiring an even higher initial chlorine dose to maintain a 5 mg/L residual. So there are two basic issues: (1) how to control the retention time, and (2) how to control chlorine chemistry.
Retention-time issues.
A properly designed chlorine contact basin must operate in a plug flow pattern to minimize short-circuiting. Retention time can be controlled by varying the liquid level so as to control the basin volume in use. This approach can only be used when sufficient head is available.
Parallel chlorine contact chamber trains add flexibility but increase project costs. They also complicate the chlorine contact basin flow splitter structure and increase head loss. Parallel trains also imply the use of more than one point of chlorine addition, although a common rapid mix chamber could be used. A single rapid-mix chamber is much easier to control and maintain. For two or more parallel contact chamber trains, each train should be built as a separate load-bearing structure.
The basin volume in operation can be controlled with manually controlled or automated slide gates at the end of each pass to link the passes in series. Shutting off the flow from one train and redirecting it to the next can control retention time. As the influent flow increases, the number of passes operated in series can be increased. This approach requires the installation of separate load-bearing walls to allow adjacent trains to be emptied.
Chlorine residual management.
Chloramination is an option for treatment plants that have a relaxed ammonia limit, i.e., a limit of more than 3 mg/L NH3-N. Chloramine is less reactive than free chlorine, and it is a good disinfectant. Adding ammonia to the WWTP effluent before adding chlorine enables use of chloramine for disinfection. Chloramine is more stable in sunlight and has fewer competing side reactions, and a lower chlorine demand will produce fewer THMs.
Meeting a CT value of 450 requires a 5 mg/L chlorine residual (0.07 millimoles of Cl2). The reaction of chlorine with nitrogen is 1 mole to 1 mole, so 0.07 millimoles of ammonia would be needed, (1.2 mg/L of ammonia or 1 mg/L of NH3-N). Allowing for some decay would necessitate as much as 2-3 mg/L of ammonia.
Upon dechlorination, chloramine will revert into ammonia. If the ammonia limit is less than 3 mg/L, it may be difficult to manage ammonia addition and avoid permit violations. Breakpoint chlorination can be used in the last pass of the chlorine contact basin to remove excess ammonia. If parallel treatment trains are used, only part of the flow needs to be treated for breakpoint chlorination; a free chlorine concentration would be maintained in a smaller portion of the basin.
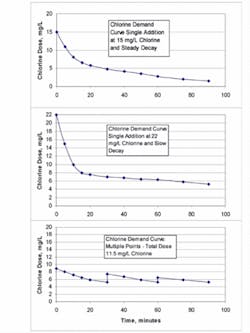
Free chlorine reactions are numerous and often depend upon the existing chlorine concentration. As the chlorine concentration is increased, more reactions or a higher chlorine demand can occur. An example of chlorine demand is shown in Figure 2.
Once chlorine demand has been met, adding more chlorine to raise its concentration to that of the initial dose may not increase the overall demand. This can be determined in the laboratory through jar testing. If the demand curve shows that a second dosing point does not exert additional demand, then adding chlorine at a second location is feasible. For disinfection systems with a long retention time, a second or a third chlorine addition point can boost the chlorine residual to meet the CT requirement.
As indicated in Figure 2, jar tests for chlorine demand must be run to simulate expected operating conditions. If the demand curve is flat after the first addition of chlorine, then several small doses of chlorine may be preferable to a single large dose. If a high chlorine dose produces a curve that is steep over the first few minutes of the reaction and then flattens out, the single dose works well. Several dosing strategies should be evaluated by jar testing to determine the best and most cost-effective approach.
Conclusions
Maintaining a free chlorine residual in a chlorine contact basin with a long retention time is difficult. For new facilities, designing chlorine contact basins for variable volume is recommended using either parallel or series configurations. These modifications will help control the actual retention time in the chlorine contact basin, and managing retention time helps manage chlorine residual.
Free chlorine typically has a faster decay rate than chloramine. Adding ammonia should be considered for chloramine disinfection if possible. However, when the effluent is dechlorinated, chloramines will be converted back to ammonia. If a plant’s discharge permit includes a strict effluent ammonia limit, breakpoint chlorination can be added to the last pass of the chlorine contact basin, either for the entire flow or only a portion of it, to manage the effluent ammonia concentration. WW
About the Authors:
Ed Kobylinski and Gary Hunter are Senior Wastewater Treatment Process Specialists in the water business of Black & Veatch, a leading global engineering, consulting and construction company. Both are members of the company’s global practice and technology team and are based in the company’s Kansas City, Mo., headquarters.