Smooth RO membrane surface decreases fouling
By: Peter Eriksson, PhD
Laboratory tests performed at Osmonics determined that its Duraslick™ reverse osmosis (RO) membrane has a lower tendency to foul than conventional polyamide RO membranes when treating surface water. Its smoother membrane surface increases fouling resistance.
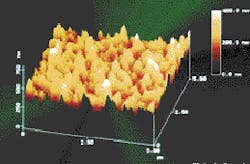
For 30 years, RO has been used in industry and municipal water systems, mostly to treat well waters. Polyamide membranes are used almost exclusively, which give the desired separation at a relatively low operating pressure, with membrane lifetime frequently exceeding five years; however surface water treatment presents different problems. Polyamide membranes frequently experience fouling problems that require monthly or more frequent cleanings. Foulants are organics that are present in surface feed water and/or products from microbiological growth in membrane elements. For this reason, membrane manufacturers have tried to modify their polyamide membranes, to decrease their fouling tendency.
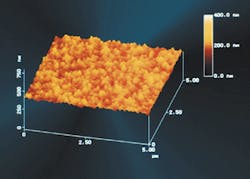
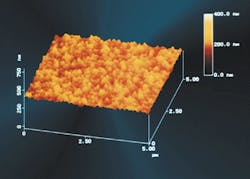
Factors that affect the fouling rate include the surface roughness of the membrane; electrostatic interaction between the membrane and the foulant; and van der Waals attraction forces. The importance of each factor in regards to membrane fouling is not fully known, but Osmonics decided to examine the effect of a smooth membrane surface when operating on natural waters. This membrane with the smooth surface, Duraslick, is an aromatic polyamide RO membrane, which is a modified DS-11, another membrane manufactured by Osmonics.
Osmonics performed a laboratory test to determine whether the Duraslick membrane had less fouling tendency than the DS-11 membrane by adding 50 mg/l of tannic acid to the tap water feed, while the pressure remained constant. Tannic acid was chosen because it is commonly present in surface waters. The temperature increased from 25ºC to 29ºC with the pure tap water feed and stayed between 28ºC and 32ºC during the remaining 24 hours of the test. The tannic acid addition decreased permeate flux by 25% for the DS-11 membrane, compared to only nine percent for the Duraslick membrane.
Patrick H. Kinghorn and William E. Haas of Ecolochem compared the Duraslick membrane with a competitor's low fouling membrane (H LFPA) and a CA membrane (H CA). A reservoir supplied the feedwater. Pretreatment consisted of clarification, multimedia filtration and acid addition. Three identical RO units in parallel each produced 200 gpm of permeate. One of the RO units was loaded with H LFPA elements, while the other two RO units were loaded with Duraslick and H CA elements, respectively. The required feed pressure is expected to be higher in winter than summer because of the temperature difference during the seasons.
The cellulose acetate (H CA) elements fouled the least followed by the Duraslick elements. Neither of those needed any cleanings. The H LFPA elements started out with 125 psig feed pressure, compared to 160 psig for the Duraslick elements, despite a lower temperature in the former case. After two months of operation, however the feed pressure for the H LFPA elements was the same as the starting pressure of the Duraslick elements. Thereafter, the required feed pressure was about the same for these two elements, with the difference that the H LFPA elements needed to be cleaned about every three months, while the Duraslick elements did not need any cleaning.
Author's note
Peter Eriksson, PhD, is the senior application development engineer for Osmonics, based in Vista, California, USA.