Water from Air
Capturing MENA's Flue Gas
Capturing water from power plant stacks and filtering it through selective hollow fibre membranes could turn water consuming power plants into water producers. Ludwin Daal provides an update on the CapWa project, including manufacturing suitable membranes.
One of the major challenges of this century is the provision of safe drinking water for a growing population, especially in Middle East regions. The shortage in water resources in arid areas requires the availability of more efficient and cheaper (drinking) water production processes.
Interestingly, air can contain large quantities of water vapour. Based on statistics provided by UNESCO, it appears that annually 575,000 cubic kilometres of liquid water evaporate and disappear into the atmosphere. This atmospheric water is much more uniformly distributed compared to any other water source. In many industrial production processes, evaporated water is present. In these processes, this water vapour is liberated or escapes as "waste" water to the atmosphere. Capturing and reusing water from industrial processes can help conserve this valuable resource. An important and localised example of these industrial processes is power generation.
For example a typical 400 MWe coal fired power plant equipped with a Flue Gas Desulphurization (FGD) unit needs about 30 m3/hour of process water. At the same time 150 m3/h of water exits the stack in its vapour form. A similar sized gas fired unit would emit between 100 – 150 m3/hour yet new gas fired units would require no more than 1 m3/hr.
Now, let's look at the installed capacity in the MENA region which in 2003 was about 178 GW (source: IEA) and which will grow to 300 GW by 2030. With 70% of this being gas fired, we are dealing with about 356 million m3 of water being emitted in 2003 and about twice as much in 2030.
To put this into perspective, the water consumption in Qatar is ~ 300 L/capita/day and in arid countries like Ethiopia approximately ~ 20L/capita/day. From these values it becomes clear that the available water potential could supply many people. Moreover, Total Dissolved Solids (TDS) levels of this evaporated water are much lower compared to surface and/or well water, with the quality of the water captured from a gas fired plant being slightly better compared to that captured in coal fired power plants.
The calculations above do not take into account the evaporated water being emitted by cooling towers, since it is difficult to determine the exact amount and type of cooling towers. That said, open system recirculating cooling towers evaporate between 0.4 and 3.8 m3 water per installed MW per hour. Moreover, aside from energy generating processes, some other industrial processes produce considerable amounts of water vapour as well, for example petrochemical industries and cement kilns.
If only the evaporated water could be captured in an energy efficient way, it could help with the daunting challenge of the water shortage in the MENA region.
A new idea is born
The idea of capturing water from flue gases did not come out of the blue. In the late nineties of the last century, energy consulting and testing & certification firm KEMA – on behalf of the Dutch power industry – carried out a lot of work in making high quality water from surface water. To achieve this, classic technologies like Ion eXchange (IX) resins as well as membrane technologies were used. At the end of the study, it appeared that surface water was a "cheap" source but that the quality of the surface water put a lot of stress on the technologies, which made the quest for alternatives relevant. A new possible water supply for the power industry then became interesting: water from their own stack. A lot of water leaves the stack and its quality is potentially far better then that of the raw surface water.
The first important question to answer, then, was how could this water be captured? Condensation of the water was not an option for several reasons. First, the use of polymer heat exchangers to condense the water out of the flue gas is quite expensive. Secondly, as soon as water starts to condense some of the other compounds present will be captured by the water as well. Thirdly, considering that flue gas contains only between 8% and 11% water vapour in a power station stack it does not really make sense to cool down the whole stream. This cooling is also not really present in typically hot and dry areas.
Gas separation membranes were considered to be the answer. In collaboration with Twente University (TU), a membrane material was developed that had a high water vapour selectivity over nitrogen. The need for a high selectivity has two major reasons. Firstly the water has to be as pure as possible and secondly the technology in mind behind the membranes does not allow any noncondensables. Nitrogen, being the main noncondensable in any flue gas, makes this selectivity need clear.
A basic, schematic depiction of how gas separation technology could be implemented in a power plant is shown in figure 1. In this situation – a coal fired power plant with FGD – the membrane modules are placed downstream of the FGD, but upstream of the flue gas reheater system. The recovered water vapour (permeate) is transported directly to the existing condenser system, where condensation takes place. From this schematic it is quite clear that the recovered water vapour should not contain any noncondensables, since this will negatively effect reliable operation of the vacuum condenser.
First prolonged field test
Several research projects, granted by the Dutch Government, have been conducted to test and develop the membrane technology. First, proof of principal tests in 2000 showed that it was possible to recover water from flue gas by means of gas separation membranes. It appeared that two main issues remained to be tackled: the selectivity of the membrane material and the improvement of the water flux.
In order to simulate normal power plant operation, even with upsets, the membranes were exposed for 24 hours a day, seven days a week for extended periods of time. The flue gas was taken from a bypass connected to the stack after and before the reheating system and was fed to a membrane chamber containing several hollow, straw-like, fibres hand-made by TU.
During the test period, several disturbances occurred, ranging from relatively high temperatures to fly ash transport caused by malfunctioning of the ESP filter. Despite these events most of the hollow fibres still had a good H2O/N2 selectivity after the test period. The target permeance value was set at >85,000 barrer and the exposed fibres showed a selectivity >100,000 barrer. However, improvements were still possible. The quality of the captured water still had to be improved if it were to be used as boiler feed water, which have conductivity levels of 0.20 µS/cm. The impurities were mainly due to sulphate, originating from the residual SO2 in the flue gas. In addition the average water flux was rather poor at 0.2 L/m2/h.
The importance of water flux improvement has to do with the fact that the membrane surface area that can be integrated in an existing power plant is limited. Take for example, the 400 MWe coal fired power plant that is required to cover its own demin water needs from captured water. At this flux an installed membrane surface area of approximately 150,000 m2 is necessary. Interesting figures perhaps for a membrane manufacturer, but it is quite a challenge for a system integrator. For this reason, further development to improve the flux was necessary.
The development route
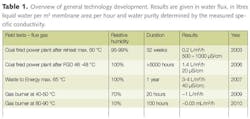
From 2004 to 2010, a combination of field tests inside flue gas streams of a coal fired power plant, a waste to energy plant and a small gas burner resulted in improvements in flux, water quality and also durability of the materials used (see also figure 2). The development and improvements led to better than expected flux results. A summary of the development route over the years is depicted in the table 1. The test results of the gas burner in 2010 show that, even in "dry" flue gas conditions, water capture is still possible. The results of the last gas burner tests (2009 & 2010) prove that one is able to capture at least 40% of the evaporated water.
Industry benefit
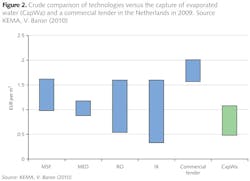
In conclusion, the ten-year research made clear that the selective hollow fibre membranes are very capable of capturing water from flue gas. At the start of the project, it was assumed that it would be possible to recover 20% of the water in flue gas, but research revealed that at least 40% could be recovered, turning a water consuming power plant into a water producer. Moreover, economic desk studies for a 400 MWe coal fired power plant showed that the water recovery process is competitive with classical IX resin, thermal distillation and membrane technologies, as shown in figure 2.
Here, the assumption is made that the price of the water capture membranes are similar to that of Reversed Osmosis membranes. It should be noted that the graph doesn't show the savings incurred if pre-heater efficiency steps are taken within the power plant, and corrosion mitigation is taken into account. Calculations show that a coal fired power plants (with cooling water access) can increase its efficiency by about 0.2% point.
Another example of savings occurred by the technology is the occurrence of Alga Bloom – Red Tide – which is a well known phenomenon in the Gulf of Oman. In Oman a bloom caused desalination plants to seize operation. Water was prioritized for drinking water purposes and power plants had been shut down. For a typical 400 MWe power plant the daily production loss would amount to EUR 1.2 million including penalty fees for not delivering. If we assume that the frequency of this bloom occurs every year with subsequent plant shut down then the value of the ultra pure water is actually 150 EUR per m3. If a shut down would occur every 19 years, the average value would be 10 EUR per m3.
The CapWa Project
The promising results cried out for a follow up. Commissioned by the European Union and led by KEMA, fourteen partners from Europe, the Middle East and Africa are now working together in an open innovation structure on a follow-up to this research. The project bears the name CapWa, 'Capture of evaporated Water with novel membranes'. The project's main goal is to develop and manufacture membrane modules, ready for industrial use within three to four years.
Within the project, the membrane modules will be demonstrated at gas and coal fired power stations, a geo-thermal well in Tunisia and paper factories in the Netherlands and South Africa. These tests should clear the way for industrial production and large-scale implementation of this new technology. The project started in late 2010 and an important milestone has been reached for the first prototype modules with manufactured membranes and selective coating havign been used. Previously the latter was always made in the lab.
Gabriel Jinjikashvily, sustainable R&D manager at the Ruthenberg coal fired power plant, where water is purchased from a water company at a rather high price: "This water is fairly clean, but needs two processing steps before we can use it in our processes. However, tests show that the water captured with the membrane technology is so clean that only one step is needed. Taking into consideration the water shortage that influences the water price, it would be economically beneficial to implement the water capture technology."
Reducing the water shortage
Results are being received from the project's first prototype membrane modules in the field. Prototypes are using commercially available materials and membranes - the results are promising.
Aside from the power industry, the technology has the potential to be applied in many different industrial processes. The ultimate choice of using the technology will not only depend on the cost of the technology but also on the industrial process, the local conditions and public opinion.
Nevertheless, the industry and, in particular the power industry, can in a few years make a valuable social contribution to the world wide water shortage, making it a better place for those who need it most.
Author's note: Ludwin Daal is a process & cooling water consultant at KEMA and project manager of CapWa. For more information on the project please visit www.watercapture.eu or email [email protected].
More Water & WasteWater International Current Issue Articles
More Water & WasteWater International Archives Issue Articles