By Brad Buecker
Primary cooling at steam generating power plants and many other heavy industries is a critical aspect of operation, and upsets can cost a great deal in lost efficiency and production. But often overlooked are auxiliary closed cooling water (CCW) systems, which also serve vital functions. Failure of a closed system has the potential to shut down a portion, if not all, of the plant. This article examines important closed cooling water treatment issues.
CCW Applications
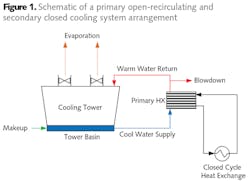
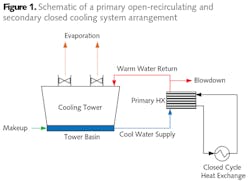
Among the equipment that may be on closed cooling networks are steel mill furnace housings, refinery and chemical plant process heat exchangers, turbine lubricating oil coolers, pump bearings, air compressor interstages, building heating/cooling systems, and more (see Fig. 1).
The aspect that makes closed cooling so suitable for many applications is that the chemistry can be carefully controlled. But, neglect of water treatment and monitoring can lead to corrosion and fouling, which may be very troublesome.
For example, a common type of closed cooler is the plate-and-frame (P&F) heat exchanger (see Fig. 2). This design maximizes contact (non-direct) between fluids but at the same time presents potential problems due to tight spacing between plates, which can become clogged with corrosion debris or microbiological slime.
Figure 2. Plate-and-frame heat exchanger.
Another common heat exchanger is the tube-and-shell type. These may not have the same tight configurations as their plate-and-frame cousins, but corrosion that causes tube failures will shut down processes equally effectively.
CCW Treatment
The term “closed” is somewhat of a misnomer, as virtually all cooling water systems have leaks or small losses somewhere that require makeup. (If serious corrosion has occurred, these losses may be significant.) Systems are often designed with a head tank for water makeup and to handle changes in demand. Thus, it is often possible for some oxygen to enter the cooling water, which, of course, can influence the corrosion potential.
Although it may be possible to utilize water with varying qualities in CCW systems, a common choice (and the main focus of this article) is condensate or demineralized water that is specially treated. Selection of condensate over less pure water minimizes the possibility of difficulties from scale-forming hardness compounds or corrosive agents such as chloride and sulfate.
Typical piping material for CCW systems is carbon steel, with stainless steel or perhaps copper alloys being a common choice for heat exchanger tubes, or plates in a P&F exchanger. Other metals may include aluminum or those metals contained in the solder of fittings within heat exchanger cooling coils. When planning a treatment program, it is important to know the entire system metallurgy.
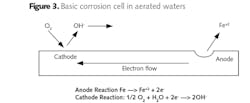
At this point, a brief review of basic corrosion is useful. All corrosion mechanisms are electrochemical in nature, although some, such as erosion corrosion, are also influenced by mechanical factors. Figure 3 offers a schematic of the primary corrosion cells that can arise in water with oxygen present.
Iron is oxidized at the anode and enters solution as the ferrous ion (Fe+2). The process releases electrons that flow through the metal to the cathode, where the electrons reduce dissolved oxygen to hydroxyl ions (OH-). Hydroxyl ions then react with the iron ions to complete the circuit and form an initial product of Fe(OH)2, which continues to oxidize to eventually form rust, with a basic formula of Fe2O3∙xH2O. Uncontrolled oxygen attack can cause severe damage in piping networks and also generate deposits that may partially or completely restrict flow.
Corrosion inhibitors function by slowing down reactions at the anode, the cathode, or sometimes both. For years during the last century, chromate (CrO42-) was very popular for corrosion control in many cooling systems, both open and closed. The ion reacts with iron to form what can be thought of as a surface layer of stainless steel, which is quite protective. However, environmental issues with regard to chromium discharge, particularly in relation to the toxicity of hexavalent chromium (Cr6+), essentially led to abandonment of this treatment scheme.
Now the most common treatment method based on cost and ability to protect carbon steel is nitrite via injection of sodium nitrite (NaNO2) to the cooling circuit. When carbon steel is first placed into service the metal surface develops an oxide layer. Although this oxide coating is in itself a function of corrosion, the layer serves as a protective film for the base metal underneath. However, the natural oxide layer can be damaged by mechanical influences or penetrated by corrosive agents.
Nitrite forms a passivating iron oxide film at the anode. Nitrite complexes with iron surfaces to form an extremely thin and highly tenacious corrosion product layer.1 The tight oxide layer limits the release of iron ions at anodic sites. A representative reaction of this chemistry is outlined below.
9Fe(OH)2 + NO2- → 3Fe3O4 + NH4+ + 2OH- + 6H2O
An important aspect to be noted in this equation is that the nitrite reaction produces ammonia, which can induce corrosion of copper alloys, particularly if an oxidizing element or compound such as oxygen is also present in the water. The pH of these solutions is typically adjusted to a range of 8.5 to 11 with an alkaline compound such as sodium hydroxide or the buffering agent sodium tetraborate, commonly known as borax.
A key concept of anodic inhibitors such as nitrite is that the chemical concentration must not be allowed to fall below a minimum value. If the level drops too low, anodes will develop in what is otherwise a large cathodic environment, establishing localized sites for very intense corrosion. Through-wall pitting is often the result.
A common range for nitrite concentration is 500 to 1,500 ppm, although I have worked with closed cooling systems in which this range could not be maintained due to significant leaks. Treatment was suspended to protect the remainder of the piping from localized corrosion. The proper answer in such cases is repair and replacement of corroded piping to return the system to “closed” status. However, plant management may not always agree with this philosophy due to cost and complexity of the task. But, another factor that makes repair the correct choice is that large leakage requires large makeup. Excessive feed of oxygen-saturated makeup propagates the corrosion illustrated in Figures 3 and 4.
Figure 4. A pipe nearly blocked by corrosion products.
A disadvantage of nitrite treatment is that the chemical serves as a nutrient for certain bacteria, such as Nitrobacter agillis, which converts nitrite into nitrate (NO3-).
A characteristic of some microorganisms is the generation of a slime layer for protection. The slime can build up and also capture particulates, which leads to reduction of water flow and the potential for under-deposit corrosion beneath the slime deposits. I once observed a nitrite-treated closed cooling water system at a large automobile assembly plant, in which microbial slime restricted flow in the small-bore cooling coils of automated welding devices. Overheating became a very problematic issue.
Also, some microorganisms, via their metabolic processes, produce acids and other harmful byproducts that can directly attack metals via the mechanism known as microbiologically induced corrosion (MIC).
Another corrosion inhibitor option, albeit more expensive, is molybdate (MoO42-), which is generated by the addition of sodium molybdate (Na2MoO4) to the cooling water. Like chromate, molybdate binds with iron to form a surface layer of ferrous molybdate (FeMoO4). This compound provides good protection, particularly against the harmful anions chloride and sulfate. A common dosage range is 200 to 1,000 ppm, with a typical recommended pH range of 9.0 to 11.0.2
Nitrite and chromate can be blended to provide a synergistic effect, where the nitrite enhances tighter molybdate bonding. Often in these cases, the control range for each chemical is slightly lower than if utilized individually.
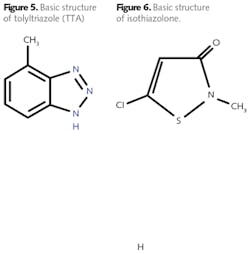
Other protection methods are available, including protection by silicates, and the use of a reducing agent such as hydrazine to maintain the passive Fe3O4 (magnetite) layer on carbon steel and cuprous oxide (Cu2O) on copper alloys. However, with regard to copper alloys most common azole chemistry is the best choice. A common member of the azole group is tolyltriazole (TTA), whose structure is shown in Fig. 5.
When utilized in basic solutions, which is common for nitrite and molybdate, the molecule deprotonates (loses the hydrogen ion), and nitrogen bonds to the copper surface. The organic rings of the compound then form a plate-like film to protect the base metal.
CCW Microbiological Control
Theoretically, in a closed system with no organic loading the conditions are unfavorable for microbiological fouling. Yet, fouling can be problematic in some systems, and particularly those that use some form of organic chemicals, e.g., azoles or dispersants, that can break down and provide food for microbes. Add a nutrient such as nitrite, or its reaction product nitrate, and microbiological growth may occur. Further, if the system utilizes water other than condensate, other microbes such as sulfate-reducing bacteria may proliferate.
Therefore, microbiological treatment may be necessary but, unlike open cooling systems, oxidizing biocides typically are not utilized in closed systems. Oxidizers can react with some corrosion inhibitors such as nitrite or introduce corrosive species, e.g., chloride, to the water. Rather, non-oxidizing biocides are the preferred choice. These chemicals function by either damaging the cell walls of microorganisms or interfering with internal metabolic processes.
Several non-oxidizing biocides are available, and this section touches upon just a few. These compounds are also utilized for biological control in open-recirculating systems.
Gluteraldehyde
Gluteraldehyde (1,5 propanedial) is a broad-spectrum biocide3 that contains only carbon, hydrogen, and oxygen. Thus, the decomposition products do not contain elements or compounds such as halides or sulfur that could cause subsequent difficulties. However, the decomposition products can potentially lower the pH of the water and provide food for microorganisms that survive.
Isothiazolone
Isothiazolone exists in several configurations with varying side groups, but the basic structure is shown in Fig. 6.
It is also a broad-spectrum biocide and is often found in household products such as liquid soap and shampoo due to its safety with regard to humans. The chemical, and stabilizing compounds included with it, will add chloride to the water. Among the problems that chloride can induce is stress corrosion cracking in stainless steel, particularly at elevated temperatures.
Dibromo-nitrilopropionamide (DBNPA)
This is a versatile biocide that is often utilized for cooling tower microbiological control and, in my experience, control of fouling in reverse osmosis (RO) membranes. Drawbacks to this compound are that it will introduce bromide ions to the cooling water and it quickly hydrolyzes at the alkaline pH in CCW circuits.
Numerous other biocides are available. A reputable water treatment vendor will be able to discuss the best approach.
Space limitations prevent a discussion of CCW systems that require freeze protection with glycol, but glycol chemistry has special features that must be considered during design and operation. Most notable is that glycol will degrade over time, and the decomposition products can seriously affect the corrosivity of the solution. IWW
About the Author: Brad Buecker is senior technical publicist with ChemTreat. He has 35 years of experience in or affiliated with the power industry, much of it in steam generation chemistry, water treatment, air quality control, and results engineering. Buecker has a B.S. in chemistry from Iowa State University with additional course work in fluid mechanics, energy and materials balances, and advanced inorganic chemistry.
References
1. Electric Power Research Institute. Closed Cooling Water Chemistry Guideline: Revision 2 to TR-107396, 2004.
2. ibid.
3. Post, R., and B. Buecker. “Power Plant Cooling Water Fundamentals,” pre-conference seminar to the 37th Annual Electric Utility Chemistry Workshop, Champaign, Ill., June 6, 2017.
Circle No. 137 on Reader Service Card