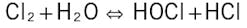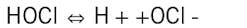Cooling Water Microbiological Control and Other Concerns
By Brad Buecker
New industrial plants are increasingly being required to use alternatives to fresh water as the source for plant makeup. One such alternative is effluent from publicly owned treatment works (POTW), also known as municipal wastewater treatment plant effluent or often simply called “gray water.” Cooling water treatment, even with fresh water as the source, can be challenging. But gray water, if not properly treated, will introduce elevated concentrations of impurities into cooling water and other process streams that can cause huge problems. This article outlines important fundamentals for cooling water microbiological control and discusses steps that can be taken to prepare gray water for use as makeup.
A Look at Fresh Water for Comparison Purposes
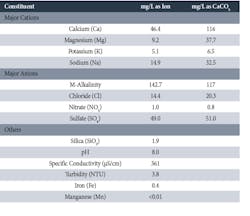
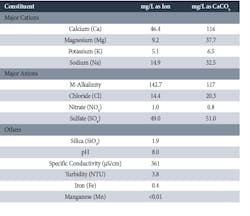
The following table is a snapshot analysis of the primary constituents from a manmade lake serving a large Midwestern power plant.
Table 1. Constituents of Typical Surface Water Supply
This water is representative, within the influences of topography, geography, and weather, of many surface supplies. The point is that control of scaling and corrosion within a cooling tower and cooling system is typically straightforward with waters of this quality. The most problematic issue, as is the case with many cooling systems, is the potential for microbiological fouling.
An Overview of Microbiological Fouling
Microbes exist everywhere in the environment and enter cooling systems with makeup water or, in the case of cooling towers, via air flow through the tower. Cooling systems provide an ideal environment, warm and wet, for microbes. Bacteria will grow in condensers and cooling tower fill, fungi on and in cooling tower wood, and algae on wetted cooling tower components exposed to sunlight. Biocide treatment is absolutely essential to maintain cooling system performance and integrity.
Bacteria are separated into the following three categories:
Aerobic: Utilize oxygen in the metabolic process.
Anaerobic: Live in oxygen-free environments and use other sources (i.e., sulfates, nitrates, or other donors) for their energy supply.
Facultative: Can live in aerobic or anaerobic environments.
A problem with microbes, particularly bacteria, is that once they settle on a surface the organisms secrete a polysaccharide layer (slime) for protection. This film then will collect silt from the water, thus growing thicker and further reducing heat transfer.
Even though the bacteria at the surface may be aerobic, the secretion layer allows anaerobic bacteria underneath to flourish. These organisms can generate acids and other harmful compounds that directly attack the metal. Microbial deposits also establish concentration cells, where the lack of oxygen underneath the deposit causes the locations to become anodic to other areas of exposed metal. Pitting is often a result, which can cause tube failure well before the life expectancy of the material.
Fig. 1. Slime and silt deposits in a heat exchanger.
Fungi will attack cooling tower wood in an irreversible manner, while algae will foul cooling tower spray decks, potentially leading to reduced performance and unsafe working locations. Cooling towers, and especially cooling tower fill, are also subject to fouling that reduces air flow, water flow, and heat transfer, and has been known to cause partial cooling tower collapse.
Thus, even with fresh water as makeup, a well-designed and controlled biocide program is essential for cooling system cleanliness. We will examine several of the most important aspects of this issue and then illustrate how POTW effluent can severely challenge such programs.
Fig. 2. Fouled cooling tower film fill.
The core of most microbiological treatment programs is feed of an oxidizing biocide to kill organisms before they can settle on surfaces. Chlorine was the workhorse for many years; when gaseous chlorine is added to water, the following reaction occurs:
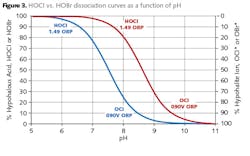
HOCl, hypochlorous acid, is the killing agent. The functionality and killing power of this compound are greatly affected by pH due to the equilibrium nature of HOCl in water.
OCl- is a much weaker biocide than HOCl, probably due to the fact that the charge on the OCl- ion does not allow it to penetrate cell walls. The killing efficiency of chlorine dramatically declines as the pH goes above 7.5. Thus, for the common alkaline scale/corrosion treatment programs, chlorine chemistry may not be efficient. Chlorine demand is further affected by ammonia or amines in the water, which react irreversibly to form the much less potent chloramines. (This is an important issue that we will re-examine when discussing POTW effluent.)
Due to safety concerns, liquid bleach (NaOCl) feed has replaced gaseous chlorine at many facilities. And some companies have developed on-site generating systems to allow production of the biocide on an as-needed basis.
A popular alternative to straight chlorine/bleach has been bromine chemistry, where a chlorine oxidizer and a bromide salt, typically sodium bromide (NaBr), are blended in a makeup water stream and injected into the cooling water. The chemistry produces hypobromous acid (HOBr), which has similar killing powers to HOCl, but functions more effectively at alkaline pH.
Another alternative, chlorine dioxide (ClO2), has found acceptance at some plants for several reasons: Its killing power is not affected by pH, the chemical does not react with ammonia, and it does not form halogenated organic compounds. Also, chlorine dioxide is more effective in attacking established bio-deposits. Drawbacks include a higher cost than other oxidizing biocides and on-site production needs (ClO2 cannot be stored).
Yet another oxidizing biocide that has proven effective in some instances is specially prepared monochloramine (NH2Cl). This compound has a much lower oxidizing potential than the biocides discussed above, but this factor appears to allow the chemical to better penetrate the protective slime layer formed by bacteria to kill the organisms underneath.
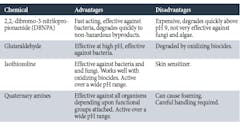
An additional method to help control microbes is a supplemental feed of a non-oxidizing biocide. Typically, feed is on a periodic but regular basis, perhaps once per week. Table 2 outlines some of the properties of the most common non-oxidizers.
Table 2. Non-Oxidizing Biocides
Careful evaluation of the microbial species in the cooling water is necessary to determine the most effective biocides. None of these chemicals should be used — or even tested — without approval from the appropriate regulating agency. They must be incorporated into the plant’s National Pollutant Discharge Elimination System (NPDES) permit. Also, as with all chemicals, safety is an absolutely critical issue when handling the non-oxidizers.
Complications from POTW Effluent
As the previous discussion hopefully illustrated, control and prevention of microbiological fouling in cooling systems takes considerable effort and operator attention, even with fresh water as the makeup. But consider if the source is secondary-treated POTW effluent. This water may contain double-digit parts-per-million (ppm) concentrations of ammonia, nitrate, organic carbon, and sometimes even phosphorus, along with substantial amounts of suspended solids. If left untreated, this “cocktail” of microbiological nutrients and food can lead to explosive growth of organisms in the cooling system.
This brings us to a key question for this article: What can be done to remove these impurities from such makeup where rapid treatment is necessary?
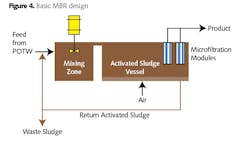
Presently, two technologies stand out as candidates for the rapid treatment needed to condition industrial plant makeup. These are membrane bioreactors (MBR) and moving-bed bioreactors (MBBR). A basic schematic of the former is outlined below.
The fundamental process is that of return activated sludge, wherein beneficial microorganisms consume the food and nutrients that enter via the mixing zone. A major differentiator of MBR from conventional activated sludge is the use of microfilter membranes rather than a traditional clarifier to separate solids from the effluent. The microfiltration process produces a very clear stream, essentially free of suspended solids.
One deficiency of this most basic MBR process is that ammonia in the stream is converted to nitrite/nitrate, but the nitrogen remains. Thus, the nitrogen can still serve as a nutrient in the plant cooling water system. This problem is solved, if necessary, by expanding the MBR system to include anoxic or anaerobic reaction chambers, with microorganisms that convert nitrite/nitrate to elemental nitrogen. The need for complete nitrogen removal rather than just a conversion from ammonia to nitrate is a decision that should be made during the design phase of the plant so that the proper MBR configuration is selected early on. MBR has also become accepted technology for raw wastewater treatment in smaller applications.
If left untreated, the “cocktail” of microbiological nutrients and food in secondary-treated POTW effluent can lead to explosive growth of organisms in the cooling system.
Regarding MBBR, the main reaction vessel includes mobile plastic media that serve as sites for the beneficial microorganisms to attach and then consume the nutrients and food from the influent.
Fig. 5. MBBR reaction vessel diagram showing mobile media. Illustration courtesy of Veolia Water Technologies.
MBBR can be likened to a very advanced version of the old trickling bed wastewater treatment process, where the beneficial microbes were attached to fixed media and then consumed nutrients and food as water flowed over and along the media. Due to the use of solid media in the reaction vessel, filtration membranes cannot be placed in this compartment. Rather, filtration must be performed separately.
MBR and MBBR Staffing
One concern often raised at industrial plants is providing staff, both from a quality and quantity standpoint, to operate these seemingly complex systems. Two ideas immediately come to mind. First, if the plant is located close to the POTW, it may be possible to place the MBR or MBBR at the POTW, and have that staff operate it. Secondly, many of the reputable manufacturers offer “build-own-operate-maintain” (BOOM) programs, where, for an annual fee, they will take care of all equipment details. IWW
About the Author: Brad Buecker recently joined ChemTreat as a senior technical publicist. He has over 35 years of experience in or affiliated with the power industry, with two additional years in the chemical process industry.
Circle No. 142 on Reader Service Card