Desalinated Water Measurement Challenges
Inductively coupled plasma mass spectroscopy (ICP-MS) is one method, says PerkinElmer, to meet tighter World Health Organization (WHO) guidance on contaminants in desalinated water for potable uses.
By Zoe Grosser
Tighter freshwater supplies coupled with growing populations have stressed conventional sources of drinking water such as ground and surface water. Groundwater pollution has further reduced sources of drinking water or increased requirements and costs for cleanup. As such, desalination is increasingly a global relief valve to these water woes. Improved technology and decreased costs make it an attractive option when compared to the rising cost of clean water obtained from other sources.
Large-scale desalination technology has been in use for decades. In fact, Aruba and Curaçao celebrated 70 years of continuous desalination operation. Over 10,000 facilities operate worldwide, with a capacity to produce more than 35 million cubic meters per day (m3/day) of fresh water. Of the 100 largest, 18 were in the USA but only one among the top 10 – and only four of the top 25 weren’t in the Mid-East. Although seawater is the primary feedstock, brackish water or treated wastewater are potential starting materials for non-coastal communities.
Supplying quality drinking water remains the primary goal of desalination. Source water and the final treated water must be analyzed to help optimize treatment and ensure drinking water regulatory requirements are met. Analytical challenges in the analysis of various types of water for desalination arise because of the matrix, which may vary and often contains high salt content.
Water Analysis Requirements
Desalinated water for drinking water and must meet regulatory requirements for microbiological, organic and inorganic contaminants. Table 1 shows of the 69 regulated primary organic and inorganic contaminants, and the maximum amounts allowed in drinking water, as prescribed in 40CFR, Part 141 of U.S. standards. Specified to measure these compounds are in the last column. Most analysis techniques have improved over the years, but ICP-MS also has been used in a new way. It and other advances are described here.
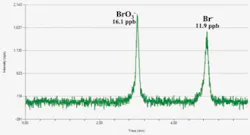
Speciated Inorganic Compounds Add to Drinking Water Characterization – Disinfection by-products (DBPs) may add to the list of measured compounds. One compound that may be observed if ozone is used for disinfection rather than chlorination is bromate (BrO3-), regulated at a maximum of 10 ng/ml. Measurement of this compound may be accomplished by measuring the total amount of bromine, and assuming it’s all present as the bromate. This approach isn’t viable, though, since bromide can be present in waters at much higher concentrations. If the limit isn’t exceeded then the total measurement is enough. But, what if the limit is exceeded? Rather than assume that the bromate isn’t in compliance, it makes sense to measure the exact compound of interest.
This measurement can be performed by two techniques coupled together (USEPA Method 321.8). High performance liquid chromatography is used for separation of bromine compounds coupled with ICP-MS for their detection. Figure 1 shows results obtained with this technique. Detection is well below compliance levels and the precision between replicates is at about 2% relative standard deviation, ensuring reliable measurements at the concentrations of interest.
Source Water and Brine a Challenge for Analysis – Source water for desalination provides a more challenging matrix for analysis, with varying salt content. Brine, a byproduct of desalination must also be evaluated before discharge, and has about twice the salinity. In the USA, metals must be measured and compared with specifications in a National Pollutant Discharge Elimination System (NPDES) permit. One example of this is measurement of trace elements, such as arsenic (As), at low levels in the presence of high concentrations of chloride in seawater. The technique with the lowest detection limit is ICP-MS, but it’s subject to interference from argon chloride (40Ar35Cl+) at the mass usually measured for As, mass 75. The sample can be diluted or an adsorbent used to separate the analyte from the matrix, but the detection limit may be degraded or losses may occur.
Newer cell technology can help remove interference before the analyte of interest gets to the mass spectrometer where it’s measured. The dynamic reaction cell (DRC) allows a gas to react with the sample and, under controlled conditions, the interference is chemically separated from the analyte. Detection limits are improved compared to other approaches and the precision in varying matrices improved. An instrument detection limit for arsenic measured in 1,000 mg/L NaCl was shown to be 2.3 ng/L, comparing favorably to detection limits of 0.6-1.8 ng/L, measured in 1% nitric acid solution, using this technique.
Conclusion
As desalination for production of drinking water increases, analytical challenges will rise also, particularly if source water. Routine analyses for source and final drinking water can be enhanced with use of new technology. Inorganic speciation analysis and development of technology to more effectively remove measurement interferences improve analyses for desalination applications.
Author’s Notes:
Zoe Grosser is a Segment Marketing Manager for PerkinElmer Life & Analytical Sciences, Contact: +1-203-402-5320 or [email protected]