Improving Effluent Treatment Plant Energy Consumption
The previous feature identified regulatory challenges to treating sewage sludge but how can upgrading an existing mechanical sedimentation treatment plant be improved? Yesterday a wastewater treatment plant, tomorrow a power plant generating clean water and recovering nutrients: how close are we? Kaj Jansson and Bengt Hansencal investigate.
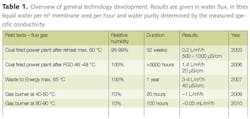
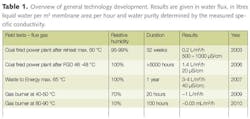
Wastewater has historically been considered as a problem and one to be disposed of. In reality wastewater contains a lot of valuable components. To demonstrate the potential values a wastewater sample has been defined in order to show how earnings/savings can be made from it. Data from several full scale wastewater treatment plants (WWTP) will be used to exemplify the theoretical part. A plant of 100,000 m3/d will be used to exemplify some parameters.
Table 1 shows a typical value of a municipal wastewater. The water itself is of value but also SS; BOD/COD, nitrogen and phosphorous is of great value if used correctly. Suspended solids consist of a lot of different components, mainly organic fibers. If these are recycled to agriculture the soil quality will be improved.
Treatment plants today and tomorrow
The wastewater treatment plant used today in many cases hardly uses all of the resources in the wastewater. A number of Nordic WWTP have been studied with mainly advanced nutrient removal and most of them could be improved. We exemplify the WWTP of today as a plant with primary sedimentation, activated sludge treatment with pre-denitrification and secondary sedimentation. The phosphorous is removed by simultaneous precipitation.
We believe the WWTP of tomorrow will take advantage of the components in influent wastewater much better than can be seen today. The plant will have a chemical pre-treatment, pre-precipitation, enhancing the separation of SS, BOD/COD and phosphorous in a primary treatment tank. This will affect the design and process choice of the secondary treatment.
If the plant has access to a cheap carbon source the natural process would be an activated sludge process with post-denitrification. This process selection is the most compact one when considering classical activated sludge processes. The process could have a small denitrification zone ahead of the aeration process, using what remains of the BOD/COD as a carbon source.
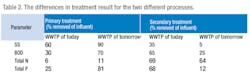
The effluent is the same as for the WWTP of today. To simplify the comparison between today's and future WWTP processes we have chosen to keep the pre-denitrification in both cases. The difference between the two processes can be seen illustrated in figures 1 and 2.
Meanwhile, Table 2 shows that the separation of different parameters change depending on process choice. Let us discuss this a little bit more in detail.
SS, BOD
With pre-precipitation a better separation of SS will cause more primary sludge. This is in general considered as a disadvantage for the pre-precipitation process but must not necessarily be, considering increased biogas potential. It must also be remembered that secondary sludge production is reduced and that primary sludge is easier to dewater, so the difference in sludge production can, in the end, be fairly small.
With a pre-precipitation process basically all particulate BOD and parts of the dissolved one is removed in the primary treatment and can directly be used for biogas production. In the WWTP of today 65% of the BOD is removed in the secondary treatment. Parts of this BOD will be biomass and parts will be removed as CO2. Potential biogas is lost.
Nitrogen
Nitrogen in wastewater mainly consists of ammonia. When ammonia is removed it is oxidized to nitrate and then denitrified. To oxidize 1 kg of NH4-N 4,56 kg of oxygen is needed, a substantial energy demand. This should be compared with energy demand when producing ammonium. 12,8 kWh (1) is used for manufacturing 1 kg of NH3-N that eventually ends up in the wastewater where most of it is denitrified to nitrogen gas and again can be used for ammonia production. The denitrification reduces the energy demand for nitrogen removal with 2,86 kg O2/kg N denitrified.
From an environmental perspective it is of course unacceptable to first spend a lot of energy to manufacture ammonia and then a lot to remove it from a wastewater. Unfortunately there are no cost efficient methods to recycle ammonia directly from wastewater. Adsorption of ammonium on zeolite is technically proven to work (3) but as long as energy and ammonia is fairly cheap this is not a viable technology.
Phosphorous
Phosphorous has a value for many agricultural purposes and should therefore be recycled. The sludge might have to be hygienized and treated before but the path to agriculture should be as straight forward as possible.
There are technologies available for recycling phosphorous, some of these have been considered as too expensive and the only one with some commercial success is stuvite precipitation (4) in reject water on WWTP using biological phosphorous removal. Then up to 50% of separated phosphorous can be separated a recycled for agricultural purposes.
Aeration Improvements
The required oxygen level in the aeration basement should be kept just above 1mg/l to save energy – more 2 mg/l has no benefit for the system and leading only to unnecessary waste of energy. Dropping the required oxygen level from 5mg/l down to 1mg/l saves around 35% of the energy required to run the aerators to the set values.
Investment in good automation improves both the process operation and the quality of the purified water. From an energy savings point of view it is hard to tell how much can be saved, but typically between 20-40% can be expected to be reached.
The importance of good mixing cannot be under estimated. A plug flow is always better than a fully mixed basin that cultivates more easily filament growth. This is because in a fully mixed basin the food concentrate drops faster, whichsupports the growth of filament because of a bigger surface area.
Biogas production
Despite its popularity across Europe, bio-P has some drawbacks such as lower biogas production, a more unstable and sensitive process and the need for coagulants to fulfill effluent standards. This is why we believe that bio-P will play a minor role in the future.
Figure 3 shows the biogas production at 33 different plants in the Nordic countries. It shows how much biogas that is produced based on the potential (BOD in influent water). It can be seen that pre-precipitation or other processes where coagulants are used is positive from a biogas perspective.
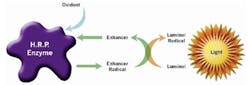
To further improve the biogas production products other than iron salts can used effectively. In a municipal WWTP, enzymatic treatment of sludge can have a positive effect. One of these products is AddZym™. This technology works in two steps with the sludge in order to increase biogas production. First an initiator is added to open up the flock structure like shown in Figure 4 (left) - an example from a full-scale plant in Sweden. Then a selected mixture of enzymes is added to the sludge supporting the hydrolysis of macro molecules like proteins and carbohydrates.
Biogas production is increased by 20%. Due to that also less organic matter ends up in digested sludge and sludge volumes decrease. On top of that the DS content in the dewatered sludge is higher, improving the sludge volume reduction even further (6).
Conclusions
Wastewater has a value that isn't fully exploited today. Energy as well of nutrients can all be recycled more efficiently. The energy balance of treatment plants can change by changing the process from simultaneous to pre-precipitation.
Therefore, WWTPs of the future will not only be facilities that remove impurities, rather facilities where valuable products can be harnessed to help fulfill the growing needs of society. The name "wastewater facility" might also have to change to reflect this evolution.
References
1.Energy Efficiency and CO2 Emissions in Ammonia Production, 2008-2009 Summary report, International Fertilizer Association
2.Matts, J. and Taylor,, A.; Rejektvattenbehandling med ammoniakavdrivning på Käppalaverket, Engineer thesis, KTH, Stockholm, Sweden
3.Ciambelli, P., Carbo, P., Porcelli, C. and Rimoli, A. (1985). Ammonia removal from wastewater by natural zeolites, I. Ammonium Ion Exchange Properties of an Italian Philipsite Tuff. Zeolites, 5, 184–187.
4.Battistoni, P.; Phosphorus removal from a real anaerobic supernatant by struvite crystallization,
Water Research, Volume 35, Issue 9, Pages 2167-2178
Author's note:Kaj Jansson is the VP, Research & Development and Bengt Hansencal is the senior project manager from Kemira.
More Water & WasteWater International Current Issue Articles
More Water & WasteWater International Archives Issue Articles