New Synthetic Media Helps Remove Iron, Manganese
Appearances can be deceiving when it comes to drinking water. While trace amounts of dissolved iron and manganese are invisible to the naked eye, the presence of these metals in small concentrations can create a variety of problems for household, commercial and industrial users.
The U.S. Environmental Protection Agency’s non-mandatory National Secondary Drinking Water (NSDW) Regulations call for dissolved iron in treated drinking water to be maintained at or below 0.3 mg/L, and dissolved manganese to be maintained at or below 0.05 mg/L. Above these threshold concentrations, iron and manganese can turn tap water rusty, create stains in laundry, and cause discoloration of porcelain sinks and tubs. If left untreated, excessive dissolved iron and manganese in water can also lead to corrosion of water-treatment equipment and distribution systems, and result in the deposition of insoluble metallic complexes inside piping and equipment components, which can reduce overall water flow through the system.
Oxidation and filtration via manganese greensand has become the most widely used method for the removal of dissolved iron and manganese in recent years because of its relative ease of operation, low maintenance, low energy requirements, and reliability. However, a prolonged shortage of greensand from its sole supplier has led to price volatility and ongoing availability problems.
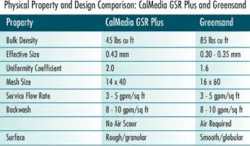
Recently, a synthetic alternative to traditional manganese greensand, dubbed CalMedia GSR Plus, has demonstrated commercial-scale efficacy for the simultaneous removal of dissolved iron and manganese during drinking water treatment. This newer oxidation-filtration media consists of granules of a proprietary, inert substrate that are coated with manganese dioxide, and offers a variety of cost and operational advantages over traditional manganese greensand.
The manganese dioxide coating on the CalMedia granules enhances the oxidation reactions that cause dissolved iron and dissolved manganese to form solid, insoluble precipitates. The coating also acts as a buffer to reduce any excess potassium permanganate (used for regeneration) in the water, ensuring that this powerful oxidation agent (and its signature purple color) does not taint the treated water or enter downstream service lines.
Similar to greensand filtration, the oxidation capacity of the CalMedia GSR Plus bed can be continuously maintained by adding a constant feed of both chlorine (fed as hypochlorite solution or in gaseous form) and potassium permanganate to the water ahead of the filtration unit. Chlorine is relatively inexpensive compared to potassium permanganate, and does most of the work converting Fe+2 to Fe+3, so when both oxidizing agents are used simultaneously, smaller amounts of the more costly potassium permanganate can be used to oxidize any remaining iron and the bulk of the dissolved manganese.
In some applications, operators may opt to regenerate the CalMedia bed intermittently, using a periodic downflow passage of a dilute potassium permanganate solution through the bed followed by a rinse cycle (such an approach typically uses a weak solution involving 1.5 to 2 ounces of KMnO4 per cubic foot of media).
Compared to manganese greensand, the granules that comprise the CalMedia GSR Plus media are larger, less dense and more lightweight. The larger particle size results in greater overall porosity and permeability of the packed beds compared to greensand beds, and this yields higher throughput rates and lower pressure drop. For non-gravity-fed systems, this can also save money by reducing pumping rates. The higher bed porosity also results in greater floc-holding capacity, which extends the filter run times between backwash cycles, and the larger particle size reduces the risk of losing smaller particles during backwashing.
An important distinction between the two competing filtration materials is that a filtration system packed with CalMedia GSR Plus requires no air-scouring step during the backwash cycle, whereas the greensand filtration system does. This simplifies the backwash cycle and elimination of the air scour step also reduces particle attrition.
Case Study
The South Bend (Ind.) Water Works supplies drinking water to 120,000 people in St. Joseph County, including 98,000 residents of the City of South Bend. The facility treats groundwater from eight different well fields. In 2004, its average production was 19.4 mgd, with a peak pumpage rate of more than 70 mgd.
An essential part of South Bend’s overall treatment train is its Pinhook filtration plant, whose gravity filters have relied on manganese greensand to remove iron and manganese from its groundwater source since a plant upgrade in 1998.
“If you’re treating groundwater anywhere in the U.S., you’re going to have a problem with iron and manganese,” said Dave Tungate, Water Quality Specialist at South Bend.
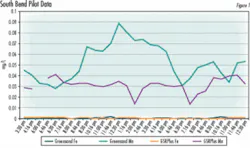
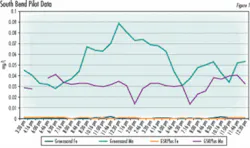
The filtration plant has a capacity of 12 mgd, and uses eight gravity filters, each of which can filter 1.5 mgd. During a routine filter maintenance overhaul in the spring of 2004, operators at South Bend found that manganese greensand levels in each of the plant’s eight gravity filters were a bit low. The utility decided to consolidate the manganese greensand in seven of its eight filtration units, and replace the oxidation-filtration media in the eighth filter with CalMedia GSR Plus, to evaluate its performance against both manganese greensand and a silica sand filtration unit it operates at another facility.
“Manganese greensand is hard to come by, and in our case, we were told that the lead time to replenish our filters would be as much as 18 months,” Tungate said. By contrast, the CalMedia GSR Plus filtration media was readily available.
After a pilot test to demonstrate that the media met the operators’ expectations, in the spring of 2004, one of the eight gravity filters at Pinhook was equipped with the new media.
At the Pinhook facility, groundwater is first dosed with chlorine and potassium permanganate and then sent through the gravity filters. The addition of KMnO4 to the water entering the filters ensures that the oxidation potential of the filtration media (both greensand and CalMedia GSR Plus) is continuously maintained. The finished water is then pumped to a storage reservoir, and either used for periodic backwashing of the filters or, with the addition of further chlorine and fluoride, is supplied to customers.
Each of the 20-ft-by-20-ft greensand filters at the Pinhook facility is configured with 18 inches of manganese greensand and a 12-inch anthracite cap. By contrast, the CalMedia filtration unit is configured with 24 inches of the filtration media and no anthracite cap.
“While the anthracite cap provides additional filtration capabilities before the water reaches the greensand layer, the light, low-density material is always at risk of getting blown out during any vigorous backwashing, so it represents an ongoing maintenance issue,” Tungate said.
Because of CalMedia’s larger overall grain size, the filtration vessel that is packed with it has a higher overall filtration rate.
“We were initially concerned that the water would not have enough retention time to ensure proper iron and manganese oxidation and removal,” Tungate said.
To compensate for this, the plant operators initially closed the CalMedia filter effluent valve partially, to slow the bed’s water-throughput rate, but they found that the Fe- and Mn-removal capabilities of the bed was comparable to the greensand vessels, even at faster throughput rates and lower retention times.
“We found that the CalMedia bed can actually filter more water than a comparably sized greensand bed and still meet our iron- and manganese-removal standards,” Tungate said.
About the Author:
Andrew McClure is the Industry Manager for the Municipal Drinking Water market at Calgon Carbon Corp. in Pittsburgh, PA. He has over 15 years experience in Applications Engineering and Marketing roles at Calgon Carbon, and is currently responsible for managing existing and new products and services that target the municipal drinking water industry. He holds a Bachelor’s Degree in Chemical Engineering from Carnegie Mellon University in Pittsburgh, PA, and a MBA from Robert Morris University in Pittsburgh, PA. He may be contacted via e-mail at [email protected].