Product Focus: Chemical Solution Helps Combat Distribution System DBPs
By October 2013, all public water utilities will be required to comply with Stage 2 Disinfection By-Product (DBP) requirements. In most cases, water leaving treatment facilities already meets those standards. However, disinfection by-products can also form when disinfectants react with scale buildup and biofilms that are often found in distribution systems.
An emerging approach to dealing with DBP formation in the distribution system is using chemical solutions to penetrate and remove the organic and inorganic deposits that react with disinfectants.
Biological and mineral deposit buildup is common in distribution systems. As scale accumulates on basin walls and pipe interiors, it becomes an excellent substrate for biofilm growth. Biofilms are covered with a slimy, polysaccharide layer that generally protects them from most primary disinfectants. These biofilms are constantly evolving and excreting organic substrates into the water surrounding the biofilm. As free chlorine reacts with these excreted organic substrates, DBPs can form.
An emerging approach to dealing with DBP formation in distribution systems is the use of NSF 60 certified chemical solutions, such as Clearitas® 101 from Blue Earth Labs. The proprietary formulation of oxidized chlorine is specifically developed to penetrate and remove the organic and inorganic deposits that react with disinfectants in the distribution system. The product is a nonhazardous chemical used online and in conjunction with traditional disinfectants.
To produce Clearitas, advanced manufacturing techniques have been developed to generate oxidants with higher oxidation states than sodium hypochlorite (NaClO). Not only are these oxidants able to dissolve different molecules present in water distribution systems, but their higher oxidation states cause them to be more hydrophobic in nature allowing them to interact more strongly with biofilm and other organics present in the water system.
The hydrophobic nature of Clearitas enables it to penetrate slime layers and break away the organic and inorganic materials in pipelines. When dosed correctly, accumulations of scale and biofilm will be broken down and dissolved back in the solution. The deposits leave the pipelines in the same manner they arrived, through the water.
Dosage rates are determined by the composition and volume of scale and biofilm within the distribution system. In heavily fouled systems, a system flushing program may be recommended to quickly remove deposits before going on a maintenance dose. Initial dosage rates range from 20 to 30 milligrams per liter (mg/l) of solution. Over time, some utilities are able to use a lower dosage rate once the distribution system is cleaned. Every system is different and must be carefully assessed to determine the cleaning process. Dosage points and levels are adjusted as field data is compiled and evaluated.
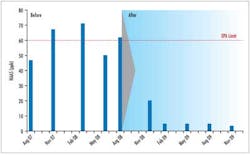
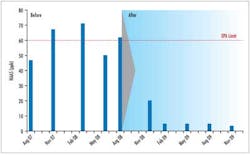
Successful applications of Clearitas have been validated at various small water systems in the U.S. Significant decreases in DBPs and increased chlorine residuals have helped the water systems meet the new Stage 2 regulations. In one study, a water utility reduced TTHMs by 55% and HAA5s by 84% over a one-year time span. Additionally, water plant personnel were able to decrease the chlorine feed by half and still maintain adequate residuals at the far ends of their system.
In another study, a utility struggling with iron deposits and loss of chlorine residuals used the chemicals to break apart the iron buildups, stabilize chlorine demand and maintain residuals in the system.