Minimizing Water Quality Impacts When Switching From Chlorine to Liquid Sodium Hypochlorite
By Kwok–Keung (Amos) Au and Jack Vogel
Many water utilities are considering switching their disinfectant/oxidant from chlorine to liquid sodium hypochlorite (NaOCl), primarily for safety and security reasons. Although both are chlorination agents, they have different impacts on the pH of the water to which they are added. pH plays an important role in the efficiencies of some water treatment processes and in the formation of disinfection byproducts (DBPs). Implementing this switch without making necessary adjustments could cause adverse effects on finished water quality.
This article provides some quantifications of the pH impacts associated with switching from chlorine to NaOCl and describes some potential side effects on treatment processes and finished water quality as a result of these pH impacts.
pH Impacts
To begin, it is important to understand the chemical reactions that occur when using these different chlorination agents.
When chlorine (Cl2) is added to water, it dissolves rapidly to form hypochlorous acid (HOCl) and reduces the pH of the water by releasing hydrogen ions (H+):
Cl 2 + H 2O ¿ HOC l + H + + Cl – (1)
HOCl is a weak acid and a portion of it dissociates to form hypochlorite ions (OCl–) and additional H+, which further decreases the pH:
HOCl = OCl– + H + (2)
When NaOCl is added to water, it dissolves rapidly to form HOCl and releases hydroxide ions (OH–), which increases the pH of the water:
NaOCl + H 2O ¿ HOCl + Na + + OH – (3)
Also, purchased NaOCl is produced at high pH, typically of a pH of 12 or higher, to maintain its stability. Thus, adding NaOCl simultaneously adds OH–.
Some of the HOCl formed from Reaction (3) will undergo the same dissociation process shown in Reaction (2) and some H+ will be released. These H+ will neutralize some of the OH– released in Reaction (3) and added with the NaOCl. Therefore, one may expect that, at the same chlorine dose, the extent of the pH increase caused by NaOCl addition will be less than the extent of the pH decrease caused by chlorine addition.
The significance of the above chemical reactions is that, unless a pH adjustment is made, the pH of water disinfected by NaOCl will be higher than the same water disinfected by an equivalent chlorine dose using chlorine. The pH difference depends primarily on the chlorine dosage and the characteristics of the water to be treated. This phenomenon is qualitatively documented in water industry literature. However, much less is known about the quantitative effects.
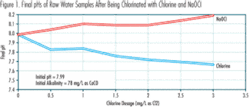
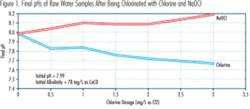
Figure 1 shows the results of some laboratory tests that were conducted at the Saginaw, Michigan, Water Treatment Plant to quantify the pH impacts caused by chlorine and NaOCl additions to a sample of the plant’s raw water. Lake Huron is the raw water source.
The raw water sample had initial pH and alkalinity values of about 8.0 and 80 mg/L as CaCO3, respectively. Chlorine solution water from the plant’s chlorinators and liquid NaOCl solution prepared in the plant’s laboratory were added to the raw water sample at various equivalent chlorine dosages. The amounts of chlorine solution water and NaOCl solution added were based on the values of their total chlorine content, measured in the laboratory immediately before their additions to the raw water sample. The final pH values, resulting from adding the two chlorination agents, were measured and are shown in Figure 1.
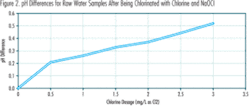
Figure 2 shows the differences in the final pH values that resulted when chlorine and NaOCl were used to achieve the same chlorine dosages. Several sets of tests were performed at this plant and the data shown here are consistent with and representative of the results of those tests.
Figures 1 and 2 indicate:
- pH increases with increasing chlorine dosage when NaOCl is used as the chlorination agent. pH decreases with increasing chlorine dosage when chlorine is used as the chlorination agent.
- At the same chlorine dosage, the pH increases caused by NaOCl addition are less than the pH decreases caused by chlorine addition. This is consistent with the fact that some of the HOCl formed by NaOCl, in accordance with Reaction (3), dissociates and releases some H+, in accordance with Reaction (2). These H+ neutralize some of the OH– formed as a result of Reaction (3) and added with the NaOCl, as described previously.
- The pH differences associated with using NaOCl instead of chlorine generally increase linearly with increasing chlorine dosage.
The impacts on pH related to adding a chlorination agent are very site–specific. Besides their dependence on chlorine dosage, the pH impacts depend on the characteristics of the water being treated. Two important characteristics are pH and alkalinity, which are related to the buffering capacity (resistance to pH change) of the water. For waters with high buffering capacities, the pH differences will be less. A utility planning a switch to NaOCl should consider running on–site, bench–scale tests to assess the potential impacts. This can be done using a jar test apparatus (as shown in Figure 3).
Potential Side Effects
The potential side effects associated with the pH differences resulting from the use of NaOCl instead of chlorine include effects on disinfection efficiency, removals of turbidity and total organic carbon (TOC), and the formations of DBPs.
- Disinfection Efficiency–
When chlorine is used as a disinfectant, the effective disinfection species are HOCl and OCl–. Their relative amounts are a function of pH; the higher the pH the less the HOCl and the more the OCl–. This is important for surface water systems because they are required to provide 0.5–log inactivation against Giardia after their treatment facilities achieve 2.5–log removal credit for this organism. HOCl is more effective against Giardia than OCl–. Therefore, the pH increase caused by switching from chlorine to NaOCl could reduce the disinfection efficiency against Giardia because less HOCl will be available. This could necessitate an increase in the regulatory C x t (chlorine residual) x (contact time) requirements for surface water systems. This is less important for groundwater systems because only virus inactivation is required for those systems. HOCl and OCl– have similar disinfection powers against viruses.
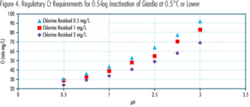
The regulatory Ct requirements for chlorination to achieve the 0.5–log Giardia inactivation at water temperatures of 0.5ºC or lower at various chlorination pH values are shown in Figure 4. These temperatures represent the worst temperature conditions in terms of Giardia disinfection efficiency. Figure 4 indicates that an increase of approximately 20% in Ct is required for every 0.5 pH unit increase. If a water utility knows the extent to which the pH will increase after the switch from chlorine to NaOCl, the site–specific impact on Ct requirements can be estimated and Ct adjustments can be planned and implemented. These could include increasing C or t, or lowering pH using an acidic chemical such as hydrochloric acid.
- Turbidity and Total Organic Carbon (TOC) Removals–
Studies have shown that when metal salts, such as ferric chloride, are used as coagulants, the removal of turbidity usually tends to improve when the pH of the water is increased. This is partly due to the fact that the quantity of metal hydroxide precipitates formed increases with increasing pH, within typical coagulation pH ranges. Therefore, at higher pHs for the same coagulant dose, more turbidity particles can be enmeshed in the increased numbers of precipitates and removed by subsequent treatment processes.
On the other hand, it is well known that the removal of TOC decreases as pH increases. This is mainly due to the fact that the negative charges carried by some of the TOC molecules increase with increasing pH, but the positively–charged products resulting from the reactions between water and metal coagulants decrease with increasing pH. Therefore, as pH is increased, the net effects are that fewer TOC molecules can be charge–neutralized by the positively charged water/coagulant reaction products and removed by subsequent treatment processes, at the same coagulant dosage.
This means that any pH increase caused by switching from chlorine to NaOCl could potentially improve the removal of turbidity, depending on site–specific conditions, but will reduce the removal of TOC. Depending on source water TOC levels, some surface water systems are required to remove certain levels of TOC. A switch in chlorination agent may, therefore, affect compliance with TOC removal requirements. Even for systems not required to remove TOC, any lowering of TOC removal would increase the potential for the formation of DBPs, due to the higher level of TOC (i.e., DBP precursors) reacting with the disinfectant.
The quantitative effect of pH change on TOC removal is site–specific. After assessing the pH difference due to a switch in chlorination agent, a water utility can decide whether an additional evaluation of the secondary impact on TOC removal is needed. This evaluation can be done through analyzing plant historical operating data and/or performing on–site testing.
- Formation of DBPs–
The formations of total trihalomethanes (TTHMs) and five haloacetic acids (HAA5s), the two groups of regulated, chlorine–related DBPs, are directly affected by pH. In general, for the same levels of TOC, chlorine dosage and contact time, the formation of TTHMs increases with increasing pH and the formation of HAA5s decreases with increasing pH. This means that a switch from chlorine to NaOCl may increase the concentrations of TTHMs and reduce the concentrations of HAA5s, due to the associated increase in pH. For example, a study has shown that when pH was increased from 7.0 to 8.0, a 20% increase in TTHMs and a 10% decrease in HAA5s were observed.
In addition to the direct pH effect on DBP formation, switching from chlorine to NaOCl also could cause indirect effects on DBP formation. As described previously, a utility may need to provide a higher Ct for microbial inactivation and may encounter a lower TOC removal, as a result of a pH increase caused by the switch. Both of these changes would increase the formation potential for DBPs due to increased amounts of disinfectant and/or disinfectant/precursor contact time being utilized and the presence of greater amounts of DBP precursors.
Like the other effects related to changing from chlorine to NaOCl, the quantitative effect of pH change on DBP formation is also site–specific. It depends on the composition of the TOC and other water chemistry factors. Utilities should consider running DBP formation potential testing to assess the effects of the pH changes, associated with the change in chlorination agent, on DBP concentrations, taking into account both the direct and indirect effects.
Concluding Remarks
- Chlorine decreases pH while NaOCl increases it.
- Laboratory testing at one water treatment plant indicates that the extent of the pH decreases caused by chlorine is higher than the extent of the pH increases caused by NaOCl. This is consistent with the chemical reactions associated with the additions of chlorine and NaOCl.
- The impacts on pH due to a switch from chlorine to NaOCl are site–specific. Chlorine dosage and the initial pH and alkalinity of the water being treated are important factors. A utility planning a switch should consider running on–site tests to assess the potential impacts.
- If it is found that this switch may cause a significant difference in pH, the potential adverse effects on treatment processes and finished water quality need to be addressed. This can be done through an analysis of the plant operating data and/or performing additional on–site testing.
- Based on the above evaluations, a utility can decide whether any process adjustments are needed such as adding more coagulant, beginning to add or adding more acidic chemical, or reducing the dosage of alkali chemicals (such as lime) currently being added to the system.
About the Authors:
Kwok–Keung (Amos) Au, Ph.D., is a Water Process Specialist and a member of Greeley and Hansen’s Water Technology Group. He specializes in water treatment processes, water quality control, regulatory compliance and water chemistry. Jack Vogel, P.E., is a Principal of Greeley and Hansen and heads its Water Technology Group. Contact Amos Au at kau@greeley–hansen.com or Jack Vogel at jvogel@greeley–hansen.com.
Acknowledgements
The authors acknowledge the funding provided by the City of Saginaw, MI, for the on–site tests and the valuable assistance provided by Amanda Kiel, Chief Chemist at the Saginaw Water Treatment Plant.